Black Headline News
LittleAfrica News
By LittleAfrica Staff
At a time when the world is experiencing an “unprecedented” number of cholera outbreaks, one of the manufacturers of the only two cholera vaccines available for use in humanitarian emergencies will cease production at the end of 2022.
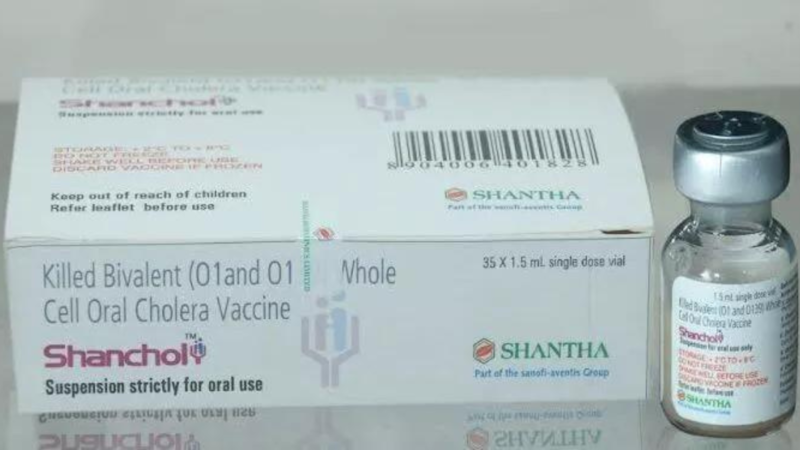
Health officials are concerned by Shantha Biotechnics’ announcement that the company will stop producing its Shanchol vaccine by the end of 2022 and stop supplying it by the end of 2023. Shantha Biotechnics is a wholly owned Indian subsidiary of the French pharmaceutical corporation Sanofi.
Only two of the three oral cholera vaccines made by EuBiologics, Shanchol, and Euvichol, have been licensed by the World Health Organization (WHO) and are accessible for widespread vaccination campaigns. These campaigns make it possible to stockpile significant amounts of the vaccine in locations where cholera is endemic and areas that may be particularly vulnerable to an outbreak during a humanitarian crisis. Shanchol comprised around 15% of the doses in the stockpile this year.
Shanchol is special in that it has been approved for use in a controlled temperature chain, allowing the vaccines to be stored for a short time under regulated circumstances at temperatures outside the typical cold chain of 2°C to 8°C.
Shantha’s decision to suspend production, according to Philippe Barboza, the WHO’s team leader for cholera, came despite repeated appeals from Dr. Tedros Adhanom Ghebreyesus, the director general of the WHO.
More Insight on the Discontinuation of Shanchol Cholera Vaccine…
According to Barboza, “To say the least, it’s a very disappointing strategy.”
The WHO estimates that cholera kills up to 143,000 people yearly in the world’s poorest nations, where access to clean water and minimal sanitary facilities is still inequitable. Currently, outbreaks are being fought in Haiti, Syria, Lebanon, Nigeria, Malawi, and Ethiopia, among other countries.
According to Sanofis’ representative, the decision to discontinue production of the vaccine was made in October 2020, and as “a responsible partner,” Sanofi “notified global health organizations and cholera stakeholders three years before supply discontinuation.”
The spokesperson stated: “We took this decision in a context where we were already producing very small volumes versus the total demand for cholera vaccines and in the knowledge that other cholera vaccine manufacturers (current and new entrants) had already announced increased supply capacity in the years to come.”
Furthermore, “the production of Shanchol will stop at the end of this year. Supply will be discontinued next year. Additionally, in the interests of the global cholera program, we entered into an agreement with our public health partners to complete a transfer of knowledge related to the manufacture of Shanchol.”
Dr. Ghebreyesus issued a warning last week that the cholera epidemic had been “turbocharged” by the climate crisis and that access to clean water has been further compromised by severe weather events including floods, cyclones, and droughts.
He emphasized that the average fatality rate from disease, which according to WHO’s data, was nearly three times higher than it had been during the previous five years this year, was of special concern.
Dr. Ghebreyesus warned that “With an increasing number of outbreaks, supply [of vaccine] cannot keep up with demand.”
He added, “We urge the world’s leading vaccine manufacturers to talk to us about how we can increase production.”
Although two doses of the oral cholera vaccine provide people with immunity for three years, it has become clear to health professionals that it is a crucial tool. Providing access to clean water and adequate sanitation remains the only long-term solution to cholera. According to Barboza, “The vaccine is the game changer because it allows countries to buy time to implement the rest.”
In order to increase supplies, Barboza is pleading with other manufacturers to step up. He added: “The makers of the other oral cholera vaccine, Euvichol, which is EuBiologics, of South Korea, were trying their best, but as you can understand, relying only on one manufacturer is extremely dangerous.”